Przecieki i osady w wymiennikach ciepła ze stali odpornej na korozję w ciepłownictwie. Zasady doboru kąpieli chemicznych do usuwania osadów z wymienników ciepła. Część 1
Types of HE used in heat engineering
Heat exchangers used in professional heat engineering are used mostly in both central heating (CH) and domestic hot water (DHW) systems. They are constructed primarily of austenitic stainless steels. Stainless steel corrosion and its basic theory were discussed in several monographs [1, 2, 3, 4]. The author of this article published before some of his experience and observations on flow and plate HE corrosion constructed of austenitic steels in 2002 [5].
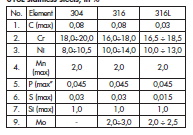
Chemical composition of stainless Cr-Ni 18-8 (ca. 18% Cr and ca. 8% Ni) steels is standardised. Their composition is determined by the European Union (EN) standards like Polish norm PN-EN 10088- 1: 1998, while many countries in the world use the standards of American Iron and Steel Institute (AISI).
–– According to the AISI standard heat exchangers are produced mainly from the following kinds of the steels:AISI 304 (equivalent of EN 1.4301 or PN 0H18N9),
–– AISI 316 (equivalent of EN 1.4401 or PN 0H17N12M2T),
–– AISI 316L (equivalent of EN 1.4404 or PN 00H17N12M2T)
Steels 304 do not contain molybdenum, while 316 and 316L steels do. Addition of titanium to 18-8 steel enables carbon increase in the steel, decreasing the steel susceptibility to intercrystalline corrosion that follows formation of chromium carbides. Molybdenum in the austenitic steels acts positively in the case of pitting and crevice corrosion caused by chlorine ions.
The chemical composition of mentioned above austenitic stainless steels is presented in the table 1.
The most popular austenitic steel HE for heat engineering are produced in two types:
–– shell and tube heat exchangers, also called flow exchangers,
–– plate exchangers in both demountable and non-demountable subtypes.
Flow heat exchangers are structures known since around 1980 as JAD and JAD-X. They are water heat exchangers, reverse-flow with diaphragm in form of tubes. Heating water flows in tubes, while heated water system flows in the shell as the reverse-flow. Advantage of these HE is their compact structure and reliable functioning, depending only on proper assembly and water treatment. Due to their structure, as a general rule, the tubes are supplied with properly-treated water, while the inner shell space is supplied with slightly treated or untreated water. The shell is an area where sediments, which should be chemically removed, are collected. Fig. 1-2 presents inlets of flow heat exchangers covered with scales, having chemical composition being mostly calcium carbonate and iron compounds.
Plate HEs reach twice as high heat transfer coefficient values compared to shell and tube types. The author presents an opinion that the plates have clean heat exchange surface as long as the scale does not cover them. These HE are constructed of thin plates made from corrosion resistant steel. Their exploitation, or chemical cleaning should not be difficult. Their compact structure causes them to take very little space. Plate HE are constructed as demountable or brazed ones. Demountable heat exchangers plates have rubber gaskets, and the whole structure is placed within a tightly joined frame. Brazed HE consist of number of plates as well. Their plates are usually brazed with copper, but joining metal may be turned to nickel or stainless steel. Internal configuration of HE channels causes the heating fluid to flow at one side of the plate, while the heated medium flows, as a reverseflow on the other side. Each HE plate has irregular surface that increases turbulence of both media, which in turn increases heat transfer between the plate and water, increasing heat transfer coefficient value.
Different producers offer various plate surfaces to create high turbulent water flow channels. Channel flow turbulence increases very much while water flow at HE inflow and exit remain moderate. Additionally it is assumed that high turbulence flow prevents or decreases scaling within the heat exchanger.
The author opinion after his participation in chemical cleaning of thousands of heat exchangers is that the smallest scaling occurred in JAD types with water free flow pass, as well as in plate heat exchangers having smooth surfaces with unobstructed flow channels, despite intense flow.
Fig. 3-4 presents plate HE covered with scale. Fig. 3 shows demountable HE, covered mostly with calcium carbonate scale, whereas Fig. 4 presents fragment of brazed HE after unaffected chemical flushing that did not remove the scale
Typical scales inside HE
Scale creation is a crucial issue as regards an effective functioning of heat engineering systems.
Scales in heat transfer installations interfere with functioning of the whole system. The scale covers the narrowest segments and may even stop work of the whole system by blocking the flow. It effects numerous unfavourable phenomena, such as significant decrease of heat amount transfer and automatic system pollution, thus causing also premature wear of water meter turbines and pump failures. Even as tiny scale thickness as 0.1 mm in plate HE may cause heat transfer to decrease by 15% up to 30%. Scaling can frequently cause the thermal and hydraulic system wrong functioning. In the CH and DHW corrosion of heat transfers installations often develops. It occurs due to the following reasons:
–– when metals having different electrochemical potentials are connected without proper insulation (macro cell corrosion),
–– at elevated temperature domestic hot water tubes corrode faster,
–– presence of microorganisms in the system, especially iron and sulphatesreducing bacteria,
–– residual oxygen dissolved in water.
Heat engineering companies have applied cleaning of HE for years by flushing with special formula chemicals to remove scales and corrosion products. The topic will be detail described in the part 2 of the paper.
There are three main kinds of scales in the heat exchangers:
a) Scales containing mainly calcium carbonate CaCO3 (Fig. 1).
They are created mostly in domestic hot water systems from water having high carbonate hardness after transformation of calcium bicarbonate into very low soluble calcium carbonate, according to the following formula:
Precipitation of CaCO3 from water having the same hardness depends strongly on temperature. During the author’s research performed for water having around 18°n (6.4 mval/l) hardness time for precipitation from water at 50°C took 1 hour, whereas at 90°C the precipitation time was only 2 – 3 seconds. Thus, it is crucial for hard water to counteract overheating by installation of effective automation. If water reaches temperature over 70°C calcium carbonate precipitation last minutes, and even the best geometrical structure of HE will not help. Precipitation in HE may potentially happen in hot water systems during thermal overheating used for disinfection to kill Legionella bacteria. In such cases chemical cleaning of HE, particularly plate ones, must be performed rather frequently, even as often as every 3 – 6 months. In the author’s opinion, standard DHW plate HE should be chemically flushed every 12 to 24 months. JAD heat exchangers used for heating drinkable water to 80°C in meat processing plants (scalding of halved pork carcasses) are regenerated chemically within time periods lasting from 2 weeks to 6 months. b) iron oxide deposits Such deposits rarely arise from austenitic steel corrosion. Original scale source for the heating side of HE is the heat transfer network, while for the heated side – the connected heat recipient system, such as CH or DHW systems. They are created as a result of carbon steel corrosion in heat transfer systems. Solid corrosion products are spread along with water through the whole system, and gather and build up usually in form of sludge or scale in HE, usually in form of magnetite.
Corrosion of steel in water systems is always initiated by oxygen (even residual) dissolved in water. Iron compounds of various oxidation level may form a protective passive layer on steel or scale on non-iron structures. Steel corrosion in waters having neutral or alkaline pH with oxygen access always occurs with oxygen depolarisation:
With oxygen access, iron (II) hydroxide Fe(OH)2 forms iron (III) hydroxide, creating oxyhydrates of goethite or lepidocrocite
At the conditions of oxygen residual amount black mixed oxide of iron (II) and iron (III) having magnetic properties is created
The above reaction occurs directly on the surface of a construction steel. Thin (several μm in thickness) and compact protective oxide layer can increase corrosion resistance of steel making it as effective as stainless steel. In this case an oxide layer will play a passive function. It grows over time but when it reaches certain thickness it is detached and enters the solution polluting the heat transfer system. In watertube boilers at higher water alkalinity or under vapour local influence (tube overheating) magnetite may be formed on the steel surface without oxygen according to the Schikorr reaction:
There is also a possibility for OH – ions transfer through external porous layer of iron (II) hydroxide, which creates conditions for magnetite creation from the precipitated Fe(OH)2 according to the Schikorr reaction:
Magnetite carried into HE from the heat transfer network or heat recipient installation causes, due to magnetic properties, further building of magnetite layers. Clogging of heat exchangers with magnetite can be limited by better corrosion prevention in heat transfer networks and internal installations, particularly the ones with untreated water. Chemical scale removal from HE of small cross section (plate HE) should be carry out frequently. Cleaning frequency depends from various factors. Due to the author experience it seems that it should be once per every 1 to 3 years. c) Scales containing calcium and iron compounds
Calcium and iron compounds containing scales are often identified in HE. They occur frequently in DHW systems if iron concentration in drinkable warm water is higher than 0.1 mg Fe/l.
Fig. 5 presents demountable plate HE with iron scale, while Fig. 6 shows flow HE inlet scaling by calcium and iron compounds.
HE leakages
Austenitic steel HE corrosion
Austenitic steel HE leakages are caused by:
a) corrosion of construction elements,
b) mechanical and other non-corrosive causes.
In JAD or JAD-X flow heat exchangers leaks happen most frequently as a result of heating pipes corrosion, particularly at connections; in DHW systems when the heated water has corrosive properties and significant amount of chlorides (> 100 mg Cl-/l). The author has not encountered HE shell corrosion during over 30 years of experience in chemical cleaning of this type of HE, which may be explained primarily by easier welding that helps to avoid corrosive changes.
Austenitic steel plate HE also corrode, albeit sporadically.
Fig. 7 presents tubes of JAD 6/50 flow HE with pitting corrosion. The HE has been previously chemically cleaned from scale with hydrochloric acid unacceptable for this type of steel. Fig. 8 presents damaged plate from demountable HE made from AISI 316 steel used in DHW system with water containing 180 mg Cl-/l. The HE was not cleaned with hydrochloric acid, however, water overheating frequently occurred as a consequence of automation failures.
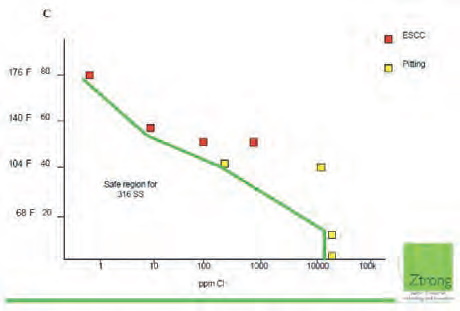
Pitting corrosion of austenitic steels, including AISI 316 steel, is strongly dependant on chlorides concentration and temperature (Fig. 9). The author knows cases of HE plates stress corrosion cracking (SCC) in the conditions of low chlorides concentration and high water temperature. Hot (80°C) drinking water containing 30 mg Cl-/l effects corrosion of AISI 316L steel plates in demountable HE (Fig. 10). The plant in which the HE was installed experienced several dozens of damaged of plates annually.
As it is shown, even usage of 316L steel does not guarantee corrosion free exploitation at high temperature and moderate amount of chlorides in water.
However, to explain pitting corrosion mechanism, occurring currently in systems constructed of 316 steel, it is not sufficient to point out thermal conditions and chloride ions concentration in heating water. As corrosive processes occur very fast (the first pits appeared as soon as within a year of usage) another factor needs to be considered. Corrosion often occurs at sensitive areas that have been formed after stamping in moulds. The other places are contact points between plates (crevice corrosion) as correct distances are not maintained because of over pressing of heat exchanger frame. The most probable scenario of 316L steel corrosion process is as follows. At temperature of 80°C and chlorides concentration of 30 mg/l, passive layer on 316L steel is locally damaged. The passive layer in the sensitive areas cannot be renewed, since they are covered by steel corrosion products. Locally, in the areas covered by thicker sediment layer, oxygen migration towards steel surface is hindered and limited. Areas of oxygen deficit appeared under the sediments due to its quick utilisation in oxidation processes. At oxygen deficit conditions, the passive layer could not be renewed. Oxygen deficit is a factor initiating activity of galvanising cells of various oxidation. Small areas of steel under sediments with oxygen deficit are anodes. The large HE surface with unlimited oxygen access works as a large cathode. Iron ions are released into the solution at anodes:
Then, as a result of iron II hydrolysis within the pits acidification starts, which additionally speed corrosion up:
At the same time oxygen dissolved in water is reduced. The reaction occurs with a simultaneous environment alkalisation:
Summary reaction will be as follows:
Further reactions are autocatalytic. Electric current running through the cells causes migration of Cl – ions into the pit and creation of concentrated solution of metal (Fe, Cr, Ni) chlorides. Metal chloride hydrolysis
causes acidification of solution in the pit and precipitation of slightly soluble metal hydroxide. Acidification increases along with increasing Cl – ions concentration speeding metal dissolution. Corrosion cell current increases effecting further transfer of Cl – ions into the pit. In accordance to the charge balance rule, anode reaction rate increase (metal dissolution) is accompanied by cathode reaction rate increase (oxygen reduction), so that all electrons released during metal ionisation may be used in reduction reaction.
Pitting corrosion process may be influenced by numerous internal (in the alloy) and external (in the environment) factors. As a general rule, the higher chrome, nickel, and molybdenum content in the steel, and the lower carbon and carbides content, secondary phases, and metallic contaminants – the lower steel susceptibility to pitting corrosion. Moreover, smooth surface is less frequently attacked by pitting corrosion than rough surface.
Corrosion is facilitated by stretching stresses resulting from metal sheets deformation during stamping, or others created during flow pulsating pressure. Since maintaining passive layer on the stainless steel is the fundamental condition for its corrosion resistance, the procedures should support its creation and renovation in the case of local damage. In order to achieve that, the following two requirements must be met:
AISI 316 steel corrosion resistance depending on chloride ions concentration and temperature (Saurce: Use of 316L in Water systems Offshore. Possibilities & Limitations, Ztrong AS, Norway)
AISI 316L corroded plate from demountable HE used for heating to 80°C drinking water containing 30 mg Cl-/l
A section of plate from copper brazed HE disassembled in nitric acid, with brazed points marked dark. Incorrectly brazed places are visible along the side wall edges
The side of plate fragment with incorrectly brazed connection points (darker points mark brazing
- maintaining clean steel surface, without any strange imbedding or polluting,
- periodical repassivation of the whole system in order to recreate the passive film at places particularly susceptible to pitting corrosion.
Both objectives may be fulfilled by periodical flushing of the system with cleaning and passivation bath, which will be described in the part 2 of the paper.
Brazed HE leakages which do not arise from corrosion
Heat exchangers producers created during the last 30 years new structures of demountable compact HE which contain large heat transfer area within a small volume. On the market, they are available as brazed plate HE. They are commonly used in Polish heat engineering for needs of both CH and DHW.
The plates are connected by welding using:
–– copper, which is the most common type of welding in the heat engineering,
–– nickel,
–– steel resistant to corrosion, usually AISI 316.
Brazed HE have undoubtedly perfect parameters within area of heat exchange. Compact structure take a little space and they are easy to assembly or disassembly. HE are equipped with stub pipes for periodical chemical flushing. High speed of flow through HE removes calcium carbonate sediments and corrosion products. It seems that plate heat exchangers are the most successful and optimal structures for the purposes of heat engineering. In the author’s opinion, if water is properly treated it does not carry solid particles of pollutions, particularly magnetite, and is not susceptible to calcium sediments precipitation, particularly calcium carbonate and calcium sulphate. Precipitation of slags, difficult to remove by chemical cleaning, creates problems for HE proper functioning. The issues include the following:
a) if the spaces between HE plates are not cleaned of slags in time than water flow is blocked (usually on the heated water side),
b) gathered sediments create perfect conditions for so-called swelling between plates and HE dimension changes, especially in areas incorrectly brazed with copper.
After numerous cleaning of HE with acidic bath, leakages mostly along external edges and between separated heating and heated medium, appeared. It was assumed that they could be a result of dissolution of thin corrosion product film with little metal. Particularities are hard to prove, when HE are non-demountable as plates are brazed with copper. The author, 15 years ago, disassembled the plates by immersion in 30% nitric acid, which effectively dissolved copper but not the plates from stainless steel. Such disassembling of the 100 kW HE lasts over one month. Disassembling of copper brazed HE may be significantly faster by cutting it into several elements and exposing to 30% nitric acid. Such procedure reduces time of HE disassembly to only a few days. Received plates may be examined visually and using microscope in order to assess possible corrosion. Plate HE elements after chemical disassembly are presented in Fig. 10.
Detailed examination showed copper brazed points in a form of characteristic “stamps” imprinted in the plate as dark grey areas, while lack of these areas indicates lack of proper brazing on shiny plate surface. The picture of the standard plate heat exchanger with incorrect brazing is presented in Fig. 11., while Fig. 12a and Fig. 12b present fragment of the same plate with imprinted “stamps and areas with no brazing.
Incorrectly Cu brazed plates may have leaking points, which are clearly visible on HE external edges. During functioning of HE a scale will gather on internal surfaces (most frequently calcium carbonate in the case of DHW). The growing scale in the areas where brazing is absent will push away the plates leading to so-called swelling. After chemical cleaning and scale removal from ‘swelled’ plates, leaks become visible mostly on the HE sides, although leaks which are generally less visible or completely invisible inside the heat exchanger are also possible. After chemical cleaning the plates do not return to their previous geometrical shape in the areas with improper brazing resulting leakages.
Low rate of HE copper brazing corrosion is also possible:
a) during normal functioning of heat exchanger in DHW system as a result of water corrosivity and turbulent water flow through plates micro spaces (low parameter),
b) at high pH > 9.2 and turbulent water flow through heat exchanger micro spaces on the side of heating medium (high parameter),
c) during or after heat exchanger chemical cleaning (this issue will be discussed in the part 2 of the paper).
Low copper corrosion rate caused by factors described as a) and b) above and high copper corrosion rate caused by incorrect chemical cleaning bath lead to loss of brazing that decides on good joining and plate stiffness. As a consequence the brazing damage leakages will exist. A question whether the plates were brazed correctly can be answered after heat exchanger chemical disassembly in nitric acid.
Copper is usually resistant against corrosive water and it cooperates well as bimetallic combination with 304 and 316 austenitic steels. The literature describes in detail corrosion of copper elements of drinkable water systems [6, 7, 8]. Copper tubes undergo specific types of corrosion, such as pitting, erosion or stress corrosion cracking. Factors influencing copper are described in PN-EN 12502-2 “Metallic materials corrosion prevention. Recommendations for corrosion risk assessment of water transfer and storage systems. Part 2. Factors influencing copper and copper alloys”.
Water in DHW system should meet at least the following conditions [8] :
– pH > 7,
– alkalinity > 2,
– ammonium ion < nitrogen 0.5 mole /l.
The standard PN–85/C0 4601 from 1985, “Water for energy purposes. Requirements and quality assessment of water for boilers and closed heat transfer systems”, still used in the heat engineering in Poland, recommends to maintain pH in 8.5 – 9.2 range for heat transfer systems with copper elements.
Copper tubes and elements in water systems may undergo erosion corrosion. It occurs when the flow becomes turbulent and protective oxide layer on copper is destroyed. Flow obtrusions, turbulences, effecting flow changes lead to transfer copper ions into the water. The literature provides various values of safe linear flow, varying from 0.5 m/s to 1.5 m/s. Obvi ously, water high corrosivity must be taken into account [8, 9]. A heat exchanger with perfectly effective heat transfer and a compact structure, may get problems of copper corrosion in micro spaces between plates in the case of the turbulent flow if water is highly corrosive against copper. However, this suggestion should be confirmed by testing dependence between copper ion concentration at the HE exit and water quality and flow rate. Besides continuous measurements of copper ion concentration, it could be useful to measure copper concentration after flow stop and restart
Conclusions
On the basis of material presented in the part 1 of the paper the following conclusions may be drawn:
A. Heat exchangers made of austenitic steel may corrode in water heating units depending on chlorides concentration and water temperature.
B. Heat exchangers leakages, particularly the ones from non-demountable plate heat exchangers, may result from corrosion but not exclusively.
C. The most frequent cause of leakages in plate heat exchangers brazed with copper is lack of the brazing. This may be a result of mistakes during assem bling or corrosion during exploitation or improper chemical cleaning of heat exchangers.
D. Checking of copper brazing is possible after chemical disassembly of HE stainless steel plates using nitric acid
REFERENCES
[1] B. Sunden, R.M. Manglik, Plate Heat Exchanger: Design, Applications and Performance, WIT Press, 2007.
[2] K. Thulukkanam, Heat Exchanger Design Handbook, Second Edition, CRC Press, 2013.
[3] A. Miguel, Heat Exchanger Failure Investigation Report, GRIN Verlag, 2014.
[4] A. Groysman, Corrosion Problems and Solutions in Oil Refining and Petrochemical Industry, Springer, 2016.
[5] J. Marjanowski, J. Ostrowski, Corrosion of Heat Exchangers Made of Chromium-Nickel Steel and Methods of Its Prevention, INSTAL, No 9, 2002.
[6] I.T. Vargas, D.T. Fischer and Others, Copper Corrosion and Biocorrosion Events in Premise Plumbing, Materials (Basel), No 10(9), 2017.
[7] J.R. Myers, A.G. Kireta, The Forms of Corrosion in Copper Tube Systems Conveying Domestic Waters, CDA Copper Symposium 2001 – Plumbing Tube.
[8] Materials of Copper Development Association Inc. , www.copper.org/enviroment/water/ NACE02122.
[9] J.M. Coyne, Flow induced failures of copper drinking water tube, Virginia Polytechnic Institute and State University, Blacksburg VA, 2009. n
Jan Marjanowski (M.Sc., Corrosion) – MARCOR Co., Gdansk